KBI has a NHP Imaging Center with wide range of the state-of-the-art technologies readily available for PD and PK with various disease models:
1.Magnetic Resonance Imaging (MRI) Platform with established methods for NASH, Parkinson’s disease, heart failure, eye disease and arthritis
2.X-Ray Platform including dual energy X-ray absorptiometry (DEXA) and X-ray digital subtraction angiography (DSA) for bone, gastric emptying, body fat/lean mass and vascular disease
3.Ultrasound Platform for heart failure, liver fibrosis, percutaneous biopsy of liver and kidney
4.Endoscopy Platform for gastrointestinal disease and gynecology disease
5.Fluorescein angiography (FA) Platform for eye disease (in Ophthalmology page).
6.Optical Coherence Tomography (OCT) Platform for eye disease (in Ophthalmology page).
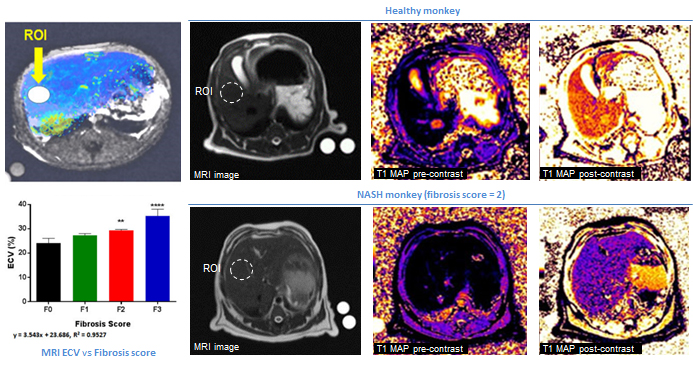
1. MRI Platform (SIEMENS 3.0T Magneton Verio)
· MRI scanning of heart and brain
· MRI quantification of liver ECV (for fibrosis)
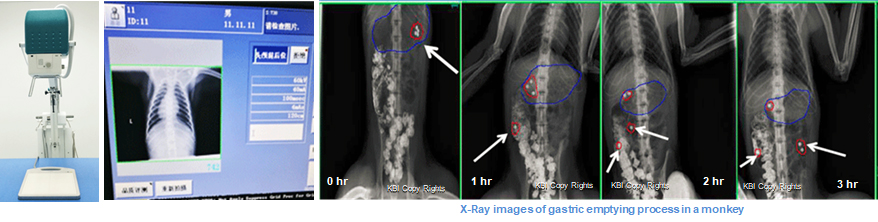
2. X-Ray Platform
· X-Ray (SEDECAL SPS-HF-4.0)
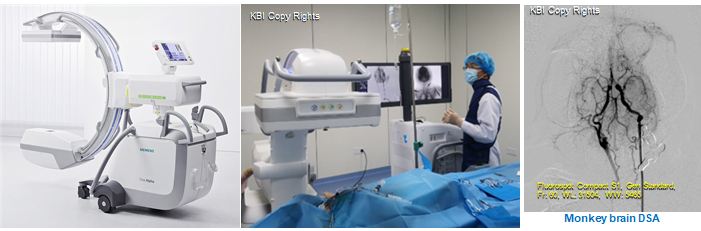
· Digital subtraction angiography (DSA, SIEMENS Cios Alpha)
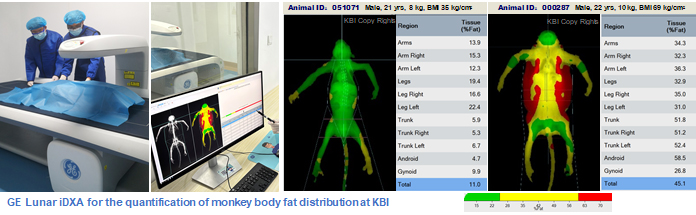
· Dual energy X-ray absorptiometry (iDXA, Discovery wi/GE Lunar 41718)
3. Ultrasound Platform (Compact console ultrasound system M9cv, ultrasound-2D echocardiography)
· Liver ultrasound images
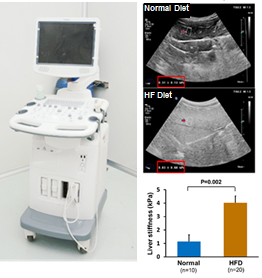
| Echocardiography video recording 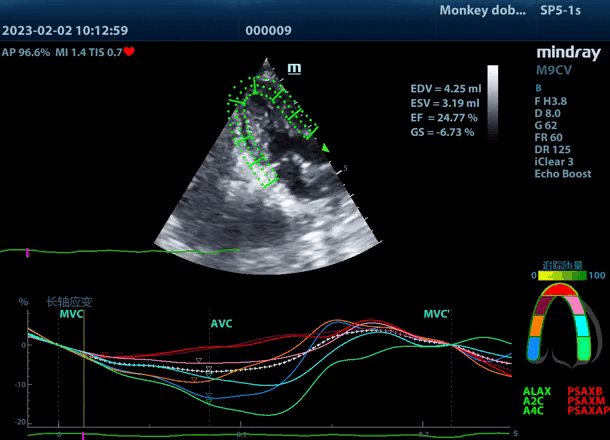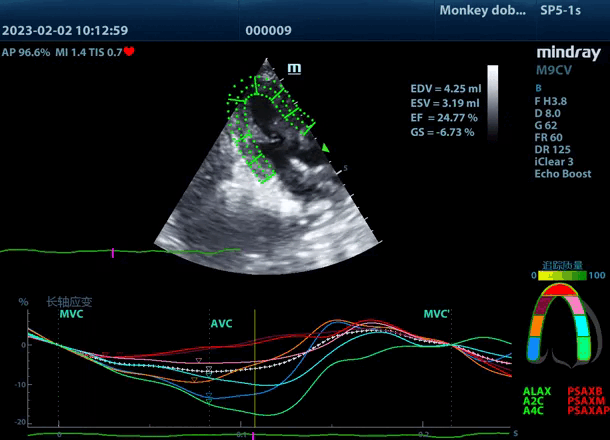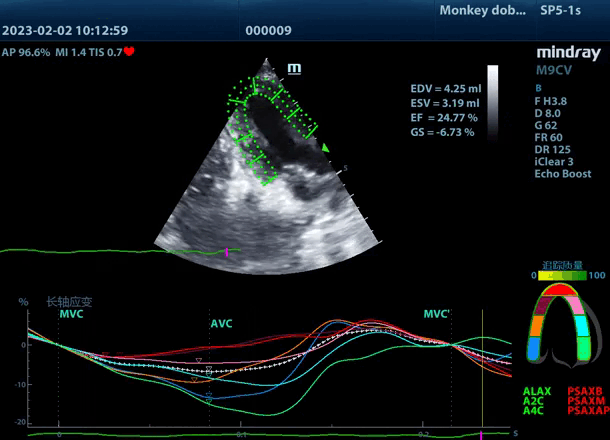
|
4. Endoscopy Platform (GV-100)
Contact us if you need more information.